Introduction: Why CE Marking Translations Matter for Global Market Access
Is your business ready to enter the European market? If so, CE Marking compliance should be at the top of your priority list.
CE Marking is a legal requirement for selling products in the European Economic Area (EEA). Without it, your products cannot legally enter this lucrative market of over 445 million consumers.
Many businesses make a critical mistake. They assume any translation will suffice for CE documentation. This assumption leads to costly compliance failures that could have been easily avoided.

Accurate translations are not just recommended—they’re a regulatory necessity. Getting them wrong can result in:
- Product recalls costing millions
- Substantial regulatory fines
- Legal penalties and market bans
- Damaged brand reputation
At its core, CE Marking indicates that a product has been assessed and meets EU safety, health, and environmental protection requirements. It’s the manufacturer’s declaration that the product complies with relevant EU directives and regulations.
The translation aspect of CE Marking is often underestimated. However, European regulations clearly mandate that all safety information, technical documentation, and user instructions must be available in the official language(s) of each member state where the product will be sold.
This requirement isn’t just bureaucratic red tape—it ensures that consumers, technicians, and regulators can understand vital product information regardless of which EU country they’re in.
Understanding CE Marking & Its Regulatory Framework
CE Marking (Conformité Européenne) acts as a passport for products entering the European Economic Area. This mandatory conformity assessment allows products to be legally marketed and sold across the EU, as well as in Iceland, Liechtenstein, and Norway.
The CE Mark indicates that your product meets all legislative requirements and can move freely throughout the European single market. Without it, customs authorities may detain your products, resulting in costly delays and potential legal issues.
1: Industries Requiring CE Compliance
CE Marking spans across numerous industries, including:
- Medical devices and healthcare equipment
- Electronics and electrical equipment
- Machinery and industrial equipment
- Construction products
- Toys and children’s products
- Personal protective equipment (PPE)
- Pressure equipment
- Automotive components
- Measuring instruments
Each of these sectors has specific requirements and directives that must be followed precisely.
2: EU Directives Governing CE Translations
The European Union has established several key directives that mandate translation requirements:
- Machinery Directive (2006/42/EC): Requires that all machinery documentation, including manuals and safety instructions, be translated into the official language(s) of the member state where the machinery is placed on the market.
- Medical Devices Regulation (MDR 2017/745): Stipulates that information supplied by the manufacturer must be in an official Union language determined by the Member State where the device is made available.
- Low Voltage Directive (2014/35/EU): Mandates that safety information and instructions must be in a language easily understood by consumers in the country where the product is sold.
General Product Safety Directive (2001/95/EC): Ensures that consumers receive all relevant information to assess potential risks throughout a product’s normal or reasonably foreseeable period of use.
According to EU regulations, the consequences of non-compliance are severe. Penalties can include:
- Immediate product withdrawal from the market
- Fines ranging from thousands to millions of euros
- Legal proceedings against company directors
- Permanent ban from selling certain products in the EU
A 2022 European Commission report revealed that approximately 32% of inspected products did not comply with EU product legislation, with documentation and labeling issues being among the top reasons for non-compliance.
Essential Documents Requiring CE Marking Translations

Ensuring proper translation of CE documentation is critical across various industries. Let’s examine the key documents requiring accurate translation and why they matter for regulatory compliance.
1: User Manuals & Instructions for Use (IFUs)
User manuals and Instructions for Use (IFUs) represent one of the most critical components of CE Marking compliance. These documents must be translated with precision into all languages of countries where your product will be sold.
Industries most affected include:
- Medical devices
- Machinery
- Consumer electronics
- Automotive
- Industrial equipment
Why accurate translations are crucial:
- They ensure end-users can safely operate products per manufacturer guidelines
- They fulfill compliance requirements under the EU Machinery Directive, Medical Devices Regulation (MDR), and Low Voltage Directive
- They prevent safety risks and potential product recalls resulting from unclear or mistranslated instructions
According to a study by the European Commission, approximately 1,000 machinery-related accidents annually can be attributed to inadequate or misunderstood operating instructions.
For medical devices, the stakes are even higher. The European Medicines Agency reports that unclear instructions contribute to approximately 7% of adverse events in medical settings.
Also read: IFU Translation: Importance, Requirements & Best Practices
2: Safety Guidelines & Technical Documentation
Safety documentation includes critical information that protects both users and operators from potential hazards. These documents must communicate warnings and procedures clearly in the local language.
Industries most affected include:
- Industrial manufacturing
- Construction Equipment
- Electrical appliances
- Engineering products
- Robotics
Why accurate translations are crucial:
- They detail hazard warnings, safety protocols, and proper installation procedures
- They ensure worker and consumer safety in high-risk industries
- They help manufacturers avoid liability claims caused by incorrectly translated safety measures
Technical errors in translated safety documentation can have severe consequences. For example, a 2018 study by the EU’s Rapid Alert System found that 23% of product recalls involving industrial equipment were linked to inadequate or incorrect safety information.
3: Declaration of Conformity (DoC)
The Declaration of Conformity (DoC) serves as the manufacturer’s official statement that their product meets all EU regulatory requirements. This legally binding document must be translated accurately.
Industries most affected include:
- All CE-marked industries
- Pharmaceuticals
- Chemical products
- Mechanical equipment
- Telecommunications
Why accurate translations are crucial:
- The DoC is a legally required document proving compliance with EU regulatory standards
- It must be translated into the official language(s) of the target EU market
- Failure to comply can result in regulatory rejection and legal consequences
The European Commission’s Blue Guide emphasizes that the DoC must be translated into the language(s) required by the Member State in which the product is placed on the market. Non-compliance with this requirement has led to products being detained at customs in multiple EU countries.
Also read: CLP Regulation EC No 1272/2008: Translation Requirements
4: Product Labels, Warnings & Compliance Markings
Product labels and warnings communicate essential safety information directly on the product itself. These must be accurately translated to ensure user safety.
Industries most affected include:
- Medical devices
- Food & beverages
- Personal protective equipment (PPE)
- Consumer goods
- Packaging
Why accurate translations are crucial:
- They help regulators, distributors, and consumers understand product safety information
- They ensure compliance with labeling regulations under EU Product Safety Directives
- They reduce risk of misuse, safety hazards, and market entry delays
The European Union Intellectual Property Office reports that incorrect labeling contributes to approximately 15% of all product compliance issues at EU borders.
5: Risk Assessments & Regulatory Reports
Risk assessment documentation outlines potential hazards associated with product use and the measures taken to mitigate those risks. These technical documents require precise translation.
Industries most affected include:
1: Aerospace
2: Automotive
3: Heavy machinery
4: Hazardous materials
5: Oil & gas
Why accurate translations are crucial:
- They outline potential safety, environmental, and operational risks of a product
- They are required for approval under EU’s risk management frameworks
- Poor translations can lead to compliance failures and operational risks
According to the European Agency for Safety and Health at Work, inadequate risk assessment documentation is cited in 28% of workplace safety investigations involving imported equipment.
Translations in compliance with CE marking requirements
The Risks of Poor CE Marking Translations
When businesses cut corners on CE Marking translations, they expose themselves to significant risks that can impact their market access, finances, and reputation. Let’s examine these risks in detail.

1: Regulatory Rejection & Market Entry Delays
Non-compliant translations can result in immediate roadblocks at EU borders. Customs authorities have the power to detain products that don’t meet documentation requirements.
- Products with incorrectly translated documentation face market withdrawal
- Manufacturers must correct violations before re-entry
- Market delays can last months, resulting in lost sales opportunities
- Distributors may cancel orders due to uncertainty
According to the European Commission’s Market Surveillance statistics, approximately 11% of products inspected at EU borders are detained due to documentation issues, including translation problems.
2: Legal Liabilities & Financial Penalties
Incorrect translations expose businesses to serious legal consequences, including:
- Regulatory fines reaching millions of euros
- Legal sanctions for non-compliance
- Product recalls and market withdrawals
- Damage to company reputation and financial standing
The European Commission actively monitors CE Marking misuse. According to EU enforcement data, authorities imposed fines exceeding €18 million in 2022 for CE compliance violations.
3: End-User Safety Risks
Perhaps the most concerning risk involves potential harm to end-users due to misunderstood instructions or safety warnings.
Real-world impact:
- Faulty translations in manuals can lead to product misuse
- Unclear safety warnings increase accident risk
- User injuries can trigger lawsuits and reputation damage
- Consumer trust is difficult to rebuild after safety incidents
4: Financial Impact of Mistranslations
The cumulative financial impact of poor translations can be devastating:
- A single mistranslation can trigger recalls costing millions
- Legal actions and settlements add significant expense
- Market access delays result in lost revenue opportunities
- Reputation damage has long-term financial implications
The European Commission estimates that the average cost of a product recall due to compliance issues exceeds €800,000, not including legal fees or lost sales.
Ensuring CE Marking Translation Accuracy

Achieving accurate CE Marking translations requires a systematic approach that combines technical expertise, quality processes, and regulatory knowledge. Here are the key strategies to ensure compliance.
1: ISO-Certified Translation Providers
Working with ISO-certified translation partners ensures that translations meet international quality standards.
Key benefits:
- ISO 17100 certification guarantees translation quality through defined processes
- ISO 9001 ensures consistent quality management systems
- ISO 13485 certification is particularly valuable for medical device documentation
- Certified providers maintain documented quality control procedures
The International Organization for Standardization emphasizes that these certifications ensure translations meet regulatory quality requirements through standardized processes and multilayer quality checks.
2: Subject-Matter Experts
Generic translators often lack the specialized knowledge needed for CE documentation. Subject-matter experts provide critical expertise.
Key benefits:
- Translators with deep knowledge of CE directives and regulations
- Industry-specific terminology expertise
- Understanding of technical concepts and equipment functionality
- Familiarity with regulatory expectations in different EU markets
Research by the European Union of Associations of Translation Companies shows that subject-matter expert translations are 78% less likely to contain critical errors in technical documentation.
3: Translation Memory (TM) & Glossaries
Translation technologies help ensure consistency across all documentation.
Key benefits:
- Ensures terminology consistency across all documents
- Creates company-specific glossaries of approved terms
- Accelerates project turnaround while maintaining quality
- Reduces cost for repeated or similar content
According to the Globalization and Localization Association, using translation memories and centralized terminology management can improve compliance document accuracy by up to 35%.
4: Linguistic & Regulatory Quality Assurance (QA)
Comprehensive quality assurance processes eliminate errors before regulatory submission.
Key benefits:
- Multi-stage review by linguistic and technical experts
- Validation against regulatory requirements
- Consistency checks across all documentation
- Final review by native speakers with industry expertise
The European Directorate for the Quality of Medicines & HealthCare recommends a minimum three-stage quality process for regulatory translations to ensure accuracy and compliance.
5: Continuous Compliance Updates
CE regulations evolve constantly, requiring ongoing updates to translated documentation.
Key benefits:
- Keeps translations aligned with new EU regulatory changes
- Updates documentation when products are modified
- Ensures continued market access as requirements evolve
- Maintains audit readiness for unexpected inspections
Global Translation Regulations Beyond CE Marking
While CE Marking is critical for the European market, businesses with global ambitions must navigate a complex landscape of international translation regulations.
1: UKCA Marking (UK)
Following Brexit, the UK introduced its own UKCA (UK Conformity Assessed) marking system.
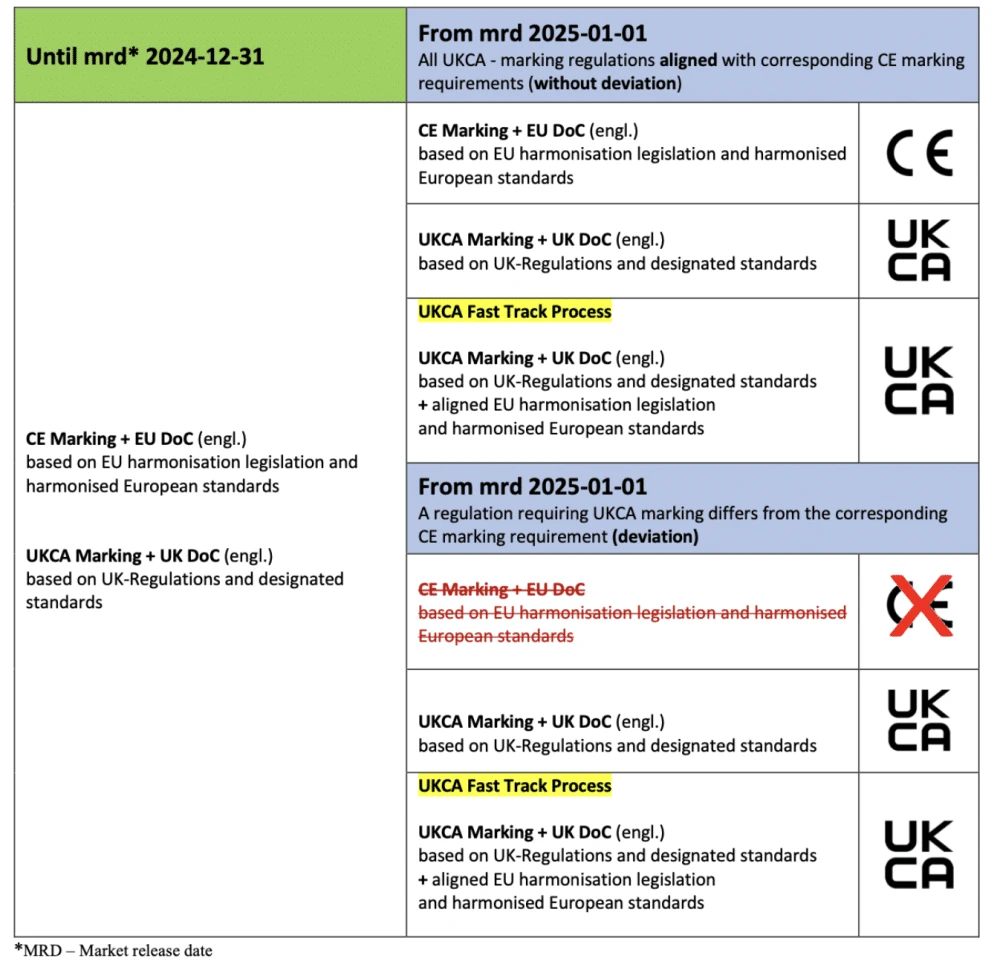
Key requirements:
- Mandatory information must be provided in English
- Welsh language requirements apply in specific contexts, particularly for public services and communications within Wales
- Technical documentation must meet UK-specific standards
- Different transition periods apply to different product categories
The UK Department for Business and Trade provides detailed guidance on UKCA marking requirements, noting that while the system parallels CE Marking, there are important differences in documentation requirements.
2: FDA Compliance (USA)
The U.S. Food and Drug Administration has its own language requirements for products sold in the American market.
Key requirements:
- The FDA mandates that all medical device labeling and documentation be in English
- Exception: Products distributed solely within Puerto Rico or U.S. territories where the predominant language is not English
- Translations must maintain the same level of detail as the original English content
- Electronic Instructions for Use (eIFU) have specific compliance requirements
According to FDA guidelines, inadequate translations in product documentation can be grounds for refusing market authorization.
3: China’s CCC Marking
China’s Compulsory Certificate (CCC) system imposes strict translation requirements.

Key requirements:
- Requires Mandarin translations for all technical documentation
- Documentation must be reviewed by authorized Chinese certification bodies
- The translation must use standardized technical terminology
- Factory inspections may include documentation review
The China Quality Certification Centre states that approximately 17% of CCC application rejections involve documentation issues, including translation problems.
4: Eurasian Economic Union (EAEU)
The EAEU, comprising Russia, Belarus, Kazakhstan, Armenia, and Kyrgyzstan, has established its own conformity marking system.
Key requirements:
- Enforces Russian translations for all product documentation
- May require additional translations into local languages of member states
- Technical regulations TR CU 004/2011 and TR CU 020/2011 specify documentation requirements
- Notarized translations may be required for certain product categories
The Eurasian Economic Commission emphasizes that documentation in Russian is mandatory, with local language requirements determined by the point of sale.
Choosing a CE Marking Translation Partner
Selecting the right translation partner is critical for CE Marking success. Here’s what to look for when making this important decision.
1: CE Compliance Expertise
Your translation partner should have deep knowledge of CE requirements and regulations.
What to look for:
- Demonstrated experience with CE-related translations
- Specialized knowledge in your specific industry
- Understanding of recent regulatory changes
- Ability to advise on country-specific requirements
According to a Nimdzi Insights report, 76% of companies that experienced regulatory issues with translations had chosen providers without specific CE Marking expertise.
2: ISO-Certified Translation Processes
Certification ensures that your translation provider follows standardized quality processes.
What to look for:
- ISO 17100 certification for translation services
- ISO 9001 for quality management systems
- Documentation of quality assurance procedures
- Regular certification audits and updates
The Association of Translation Companies recommends working exclusively with ISO-certified providers for regulatory translations to ensure error-free, regulator-approved results.
3: Multi-Language Support
Comprehensive language coverage is essential for businesses targeting multiple EU markets.
What to look for:
- Coverage of all 24 official EU languages
- Native speakers for each target language
- Cultural adaptation capabilities
- Consistent quality across all languages
4: Technology and Security Capabilities
Modern translation providers should have robust technology and security measures.
What to look for:
- Secure handling of confidential documentation
- Cloud-based translation management systems
- Backup and disaster recovery procedures
- Data protection compliance (GDPR, etc.)
Conclusion: Ensuring Global Compliance Success
Ensure CE Marking compliance with certified translations that meet the highest regulatory standards. Talk to our experts today to learn how we can support your global expansion with accurate, compliant translations for CE Marking and beyond!
Would you like to learn more about how our specialized translation services can help your business achieve CE Marking compliance? Contact our regulatory translation experts today for a consultation.
Looking for Professional Ce Marking Translation services ?
FAQS
What is CE marking and why is it important for global businesses?
CE marking is a certification that indicates a product meets EU safety, health, and environmental requirements. It is mandatory for many products sold in the European Economic Area (EEA), ensuring legal market access.
Which documents need translation for CE marking compliance?
Key documents include user manuals, safety instructions, technical documentation, packaging labels, declarations of conformity, and risk assessments. These must be translated into the official languages of the EU countries where the product is marketed.
Who is responsible for ensuring translations meet CE marking requirements?
The manufacturer or the authorized representative placing the product on the EU market is responsible for ensuring translations are accurate, complete, and compliant with local regulations.
Do all EU countries require translations in their official language?
Yes, most EU member states require product information and safety instructions to be available in their national language(s). Requirements vary, so businesses must check the specific rules for each target market.
What should I look for in a translation partner for CE compliance?
Choose a translation company with ISO 17100 certification, native industry experts, experience with EU directives, and the ability to provide certified translations when required.
What is CE marking?
CE marking is a certification symbol that shows a product complies with the European Union’s health, safety, and environmental protection standards. It is mandatory for many products sold within the European Economic Area (EEA). By affixing the CE mark, manufacturers declare that their product meets all relevant EU directives and regulations, allowing free movement of goods across EU member states.

