For pharmaceutical companies entering the Swiss market, complying with Switzerland’s regulatory framework is essential to ensure patient safety, product quality, and market access.
Swissmedic, as the national authority that governs all medicinal products through the Therapeutic Products Act (TPA), sets requirements for licensing, labelling, and marketing of all medicinal products in the region.
An important aspect of this regulation is clear and accurate communication, ensuring that healthcare professionals and patients can clearly understand drug information and use medicines safely.
This blog highlights these requirements in detail, providing insights into what needs to be translated, the responsibilities of manufacturers, and best practices for ensuring compliance.
What is the Therapeutic Products Act?
The Therapeutic Products Act (TPA), or Heilmittelgesetz (HMG), is Switzerland’s primary law governing all therapeutic products, including medicines, biologics, and medical devices. In force since January 1, 2002, it forms the foundation of the Swiss regulatory system, ensuring that any product placed on the market is safe, effective, and high quality.
The TPA defines the rules for product authorisation, market surveillance, company licensing, advertising standards, and compliance enforcement. In essence, the TPA is the legal framework that empowers Swissmedic to evaluate, approve, monitor, and regulate therapeutic products in Switzerland.
Overview of Switzerland’s Medicinal Product Regulatory Framework
In Switzerland, medicinal products are regulated under a legal framework that sets out the rules for authorisation, licensing, monitoring, and regulatory oversight. These requirements are further detailed through supporting ordinances that guide compliance throughout the product lifecycle.

1: Medicinal Products Licensing Ordinance (MPLO / AMBV)
MPLO (SR 812.212.1) regulates the licensing of companies and establishments involved in the manufacturing, importing, exporting, and wholesaling of medicinal products.
It defines the requirements for compliance with Good Manufacturing Practice (GMP) and Good Distribution Practice (GDP), ensuring consistent product quality, patient safety, and the integrity of the pharmaceutical supply chain throughout all stages of distribution.
2: Ordinance on the Requirements for the Authorisation of Medicinal Products (AMZV / OEMéd)
OEMed (SR 812.212.22): Covers product authorisation requirements, specifying the scientific and technical documentation that applicants must submit to Swissmedic to demonstrate quality, safety, and efficacy. It defines the structure of the application dossier (e.g., CTD format) and required data, including quality reports, non-clinical studies, and clinical trial results, forming the basis for marketing authorisation.
3: Ordinance on Advertising Medicinal Products (OMPA / AWV)
OMPA (SR 812.212.5): Regulates advertising and promotional activities, ensuring all claims are factual, responsible, and compliant with consumer protection standards. Strictly prohibits advertising of prescription-only medicines to the public and ensures promotional communication does not encourage inappropriate or excessive use.
4: Ordinance on Medicinal Products (OMéd / VAM)
Focus & Scope (SR 812.212.21): Provides general operational rules and market surveillance, covering dispensing categories (A, B, D, E), pharmacovigilance, storage, and ongoing monitoring. Serves as the operational backbone of Swissmedic’s oversight, ensuring safe handling, distribution, and supervision of medicinal products throughout Switzerland.
5: Ordinance on the Simplified Licensing of Therapeutic Products (TPLO / OASMed)
TPLO establishes simplified licensing procedures for certain categories of therapeutic products under defined conditions, complementing the main TPA framework.
Rules Non-Swiss Pharmaceutical Companies Must Follow to Register Drugs in Switzerland

1: Local Presence
Non-Swiss pharmaceutical companies must appoint a legally recognised Swiss entity or representative to act as the applicant or Marketing Authorisation Holder (MAH) proxy.
This ensures a responsible party is established in Switzerland to manage regulatory communication, fulfil legal duties, and respond promptly to Swissmedic throughout the evaluation and post-approval phases.
2: Quality, Safety & Efficacy Requirements
Companies must submit a complete eCTD dossier containing all scientific evidence needed to demonstrate the medicine’s quality, non-clinical safety, and clinical efficacy.
Swissmedic follows ICH guidelines and may rely on recognised foreign inspections and assessments under Mutual Recognition Agreements, helping streamline approval for globally regulated products.
3: GMP Compliance & Responsible Person
All foreign manufacturing sites must comply with PIC/S GMP standards. Applicants must provide valid GMP certificates and a declaration from the Responsible Person confirming compliance. Swissmedic may also conduct on-site inspections abroad if required to verify product quality and safety.
4: Post-Authorisation Obligations
After approval, companies must operate a Swiss-compliant pharmacovigilance system, maintain a local QPPV, report safety findings, and submit PSURs or PBRERs as scheduled by Swissmedic.
Overview of languages in Switzerland
Switzerland’s multilingual nature influences communication, education, and daily life, making language a key consideration for businesses and organizations operating in the country. Understanding and addressing this diversity is essential for effective engagement, compliance, and market success.
Official Languages of Switzerland
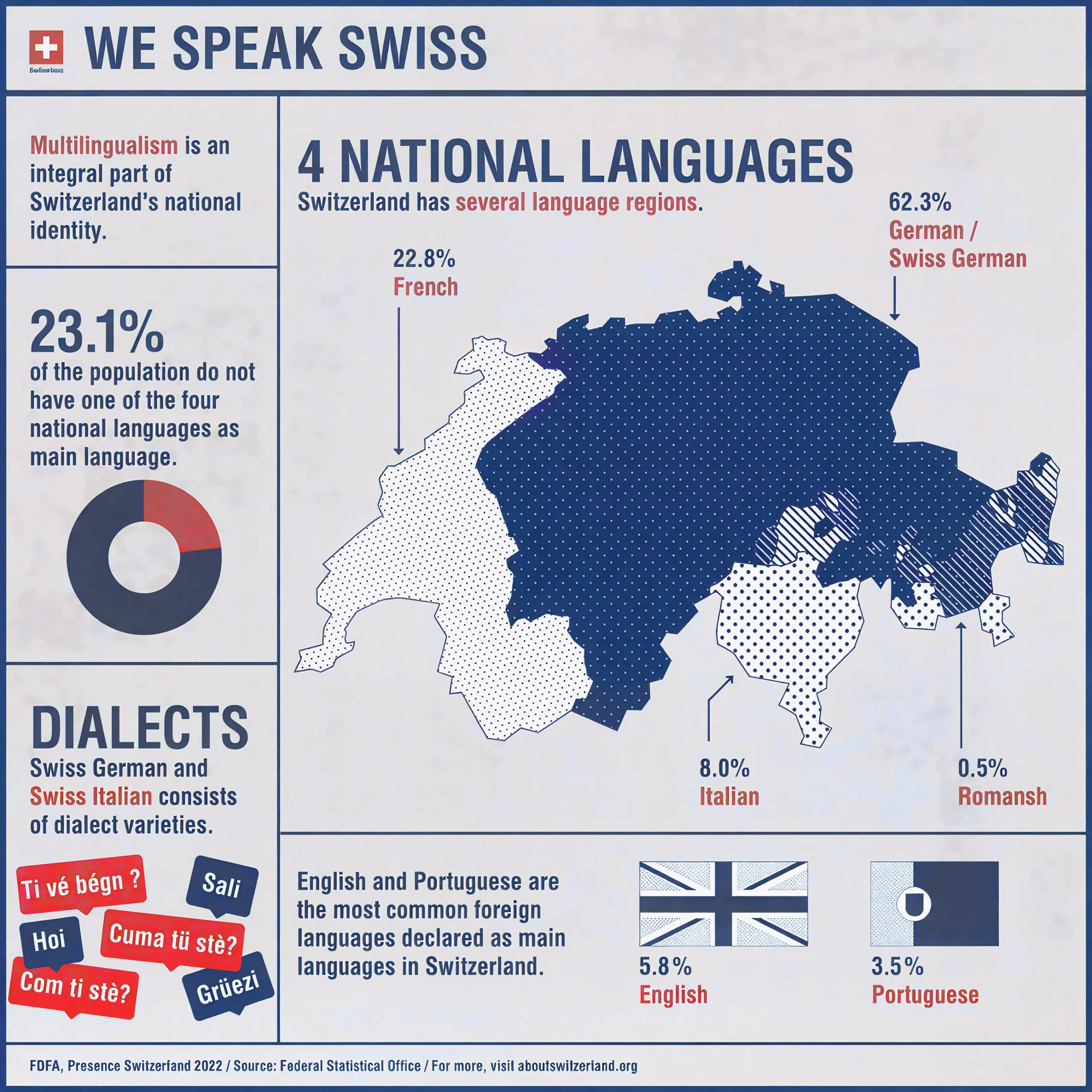
1: German
German is spoken by about 63% of the population and is widely used in northern, central, and eastern Switzerland.
2: French
Approximately 23% of Swiss residents speak French, mainly in the western cantons such as Geneva, Vaud, Neuchâtel, and parts of Bern and Fribourg.
3: Italian
Italian is spoken by around 8% of the population, primarily in Ticino and some areas of Graubünden.
Also Read: Top Languages Spoken In Switzerland
Looking for Pharmaceutical Translation services?
Language and Translation Requirements for Medicinal Products in Switzerland
1: Summary of Product Characteristics (SmPC)
The SmPC must be translated into the official language(s) of the region where the medicinal product is marketed. The translation must exactly match the content of the approved dossier to ensure accuracy and consistency for regulatory compliance and proper use.
2: Patient Information Leaflet (PIL)
The patient information leaflet must also be provided in the official language(s) of the region. The translation should be clear, easy to understand, and faithful to the SmPC to ensure patients receive correct and comprehensible instructions about the medicinal product.
Also read: Translating Patient Information Leaflets: Challenges and Solutions
3: Labelling and Packaging
Labelling and packaging can be multilingual, but they must include all official languages of the region in which the product is marketed. Multiple languages may appear on the same package or on an insert inside the packaging, as long as the information is complete, accurate, and legible.
4: eCTD Regulatory Submission Dossier
The electronic Common Technical Document (eCTD) submitted for marketing authorisation allows more flexibility in language while following international standards.
- Module 1 (Administrative & Regional Information): Includes cover letters, application forms, Swiss-specific documents, and pricing information. Typically submitted in one Swiss official language (DE, FR, or IT). Final SmPC, PL, and packaging mock-ups must still be tri-lingual.
- Module 2 (Summaries): Quality, non-clinical, and clinical overviews and summaries. Primarily submitted in English, aligning with international standards.
- Module 3 (Quality): Manufacturing, control, and stability data for drug substances and products. Submitted mainly in English, though DE, FR, or IT is also accepted.
- Module 4 (Non-Clinical): Pharmacology and toxicology study reports, generally in English.
- Module 5 (Clinical): Clinical study reports and literature references, generally submitted in English to maintain international alignment.
5: Advertising and Promotional Materials
All advertising and promotional materials must be provided in the official language(s) of the target region. The translations must maintain scientific accuracy, be factual, and comply with the Medicinal Products Advertising Ordinance (OMPA/AWV) to prevent misleading claims.
Language Rules for Healthcare-Professional vs Patient-Facing Drug Information
| Category | Document | Language Requirements | Notes |
|---|---|---|---|
| Patient-Facing (Trilingual) | Patient Information Leaflet (PIL) | DE, FR, IT | Supplied with the product or electronically. Must be clear and easy to understand. |
| Packaging & Labelling | DE, FR, IT | Essential details must appear in all three languages. Hospital-use products may use one official language or English. | |
| HCP / Regulatory | Information for Healthcare Professionals (SmPC / IHP) | DE, FR, IT | Versions must align. German is authoritative. Digitally published. |
| Package Leaflet (HCP Use Only) | DE, FR, IT | Printed PIL may be waived for HCP-only products; digital version still required. | |
| Regulatory Dossier (CTD 1–5) | EN, DE, FR, IT | English preferred for scientific content. | |
| Post-Authorisation Communications | EN or DE, FR, IT | Follow the language of Swissmedic’s request. |
Best Practices for Pharmaceutical Translation

1: Work with Certified Pharmaceutical Translation Agencies
Work with translation agencies that have ISO 17100 certification for translation quality and ISO 9001 certification for quality management. These agencies work with professional translators and subject matter experts (SMEs) who specialize in pharmaceutical terminology, regulatory documents, and drug labeling.
They ensure that complex scientific content is translated accurately, clearly, and ensure compliance with Swissmedic requirements, supporting both regulatory approval and patient safety.
2: Ensure Regulatory Compliance
Translations must strictly adhere to Swiss pharmaceutical regulations, all translations of SmPC/IHP, PIL, packaging, and labelling must convey warnings, instructions, and technical details accurately to ensure patient safety and legal compliance.
3: Use a Standardized Translation Workflow
Implement a structured translation workflow with Translation Management Systems (TMS), translation memories, and approved terminology databases. This ensures consistency across all documents, minimizes errors, and speeds up the update process for regulatory submissions or product information changes.
4: Implement Rigorous Quality Assurance
Establish multi-stage quality checks including linguistic review by native speakers, technical review by pharmaceutical experts, and regulatory audits. This guarantees that the translations meet both scientific accuracy and compliance standards.
5: Documentation & Record Keeping
Maintain comprehensive records of all translations, review checklists, and version histories. This facilitates ongoing compliance with regulatory updates, supports inspections by authorities, and ensures traceability for pharmacovigilance and quality management purposes.
Also read: Pharmaceutical Translation: Importance, Challenges & Best Practices
Milestone Localization as a Trusted Partner for Pharmaceutical Translations
We help pharmaceutical companies comply with Swissmedic language requirements, ensuring that all essential medicinal product documents are accurately translated into the required languages.
As an ISO 9001 and ISO 17100 certified company, we ensure that all translations meet strict pharmaceutical quality management standards and regulatory compliance requirements.
We provide translations into German, French, English and Italian. Our pharmaceutical translators have deep expertise in regulatory frameworks, clinical data, SmPCs, PILs, and labeling, ensuring your documents are fully accepted by Swiss authorities.
Conclusion
Compliance with Switzerland’s pharmaceutical regulations is essential for ensuring the safety, quality, and transparency of medicinal products.
Pharmaceutical companies must ensure all information for patients, healthcare professionals, and authorities is accurate, consistent, and available in Switzerland’s official languages.
Meeting these language and communication requirements improves patient safety and supports clear, informed decision-making across the healthcare system.
Also read: Swissmedic Language Requirements for Medical Device Documentation
Are You Looking for Certified Pharmaceutical Translation services?
FAQs
What is the Therapeutic Products Act (TPA/HMG)?
The Therapeutic Products Act (TPA/HMG) is the main law governing medicines in Switzerland. It sets the rules for authorisation, licensing, monitoring, and market supervision of medicinal products.
What is Swissmedic’s role in regulating medicines?
Swissmedic is the Swiss authority responsible for approving medicinal products and monitoring their safety, quality, and efficacy throughout their lifecycle.
Which languages are required for patient-facing drug information in Switzerland?
Patient-facing documents must be available in German, French, and Italian. This includes patient information leaflets and essential packaging details.
Are healthcare professional documents required in multiple languages?
Yes. Information for healthcare professionals, such as SmPCs, must be provided in German, French, and Italian, with consistent content across all versions.
Can regulatory submissions be made in English?
Yes. Scientific and administrative documents, including CTD Modules 1–5, can be submitted in English, which is commonly accepted by Swissmedic.




