For medical device and In Vitro Diagnostics manufacturers entering the Swiss market, complying with Switzerland’s regulatory framework is essential for patient safety, quality, and market access. After Switzerland’s separation from the EU system, Swissmedic introduced its own requirements through the MedDO and IvDO.
A key part of these rules is clear and accessible communication so that patients, professionals, and authorities can understand device information and use it safely.
This blog highlights these requirements in detail, providing insights into what needs to be translated, the responsibilities of manufacturers, and best practices for ensuring compliance.
What is Swissmedic?
Swissmedic is Switzerland’s national authority responsible for regulating medicines and medical devices. It ensures that all therapeutic products sold or used in the country meet strict standards of quality, safety, and effectiveness.
Swissmedic authorises new medicines, approves clinical trials, and issues licences for manufacturers and distributors. It also monitors products already on the market by investigating safety incidents, inspecting companies, and taking action against illegal or unsafe products.
Overview of Switzerland’s Medical Device Regulation and In Vitro Device Regulation
The Swiss regulatory framework for medical devices is defined by two primary ordinances, the Medical Devices Ordinance (MedDO 2002) and the In Vitro Diagnostic Medical Devices Ordinance (IvDO 2022).
A: Medical Device Regulation In Switzerland (MedDO)
The Medical Devices Ordinance (MedDO, SR 812.213), which came into force on January 1, 2002, governs the safety, performance, and market access of medical devices in Switzerland. Based on the Therapeutic Products Act (TPA, SR 812.21), it aligns with the EU MDR but operates independently, as Switzerland is no longer part of the EU’s mutual recognition system.
The MedDO sets rules for device classification, conformity assessment, technical documentation, post-market surveillance, and defines responsibilities for manufacturers, importers, authorised representatives, and distributors.
It also requires registration of devices and economic operators in Swissdamed traceability and regulatory oversight. Swissmedic enforces these requirements to ensure patient safety, market transparency, and high quality standards for all medical devices in Switzerland.
B: In Vitro Diagnostic Device Regulation (IvDO)
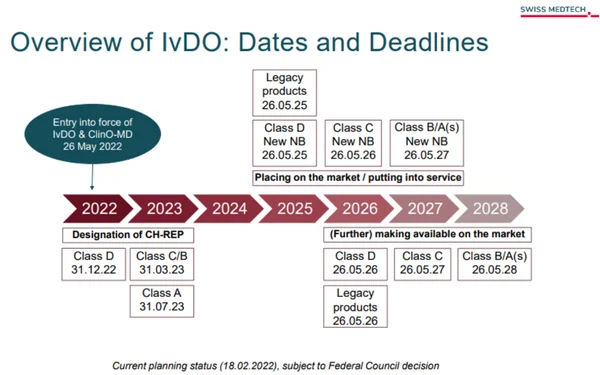
The In Vitro Diagnostic Medical Devices Ordinance (IvDO, SR 812.219) is Switzerland’s specific legal framework regulating IVD devices. It is the implementing act for IVDs under the Therapeutic Products Act (TPA) and came into force on May 26, 2022, aligning with the EU In Vitro Diagnostic Regulation (EU IVDR).
The IvDO ensures the safety, performance, and transparency of diagnostic products on the Swiss market by mandating high-quality standards and introducing a comprehensive life-cycle approach to regulation.
What are the rules that non-Swiss manufacturers should follow to register their devices in Switzerland?
1: Appoint a Swiss Authorized Representative (CH-REP)
Foreign manufacturers must appoint a CH-REP in Switzerland as the liaison with Swissmedic. The device label must include the CH-REP’s name, trade name, and address. The CH-REP verifies compliance, retains documentation, manages incident reporting, and cooperates with Swissmedic.
2: Register in Swissdamed (CHRN & Devices)
Swiss-based CH-REPs and importers must register to obtain a CHRN (mandatory since August 2024). Device/UDI registration is voluntary now and becomes mandatory on July 1, 2026. Devices involved in incidents or recalls must be registered immediately.
3: Importer Labelling
The Swiss importer’s name, trade name, and address must appear on the device or packaging. Importers verify CH-REP validity and compliance before market placement.
Overview of languages in Switzerland
Switzerland’s multilingual nature influences communication, education, and daily life, making language a key consideration for businesses and organizations operating in the country. Understanding and addressing this diversity is essential for effective engagement, compliance, and market success.
Official Languages of Switzerland
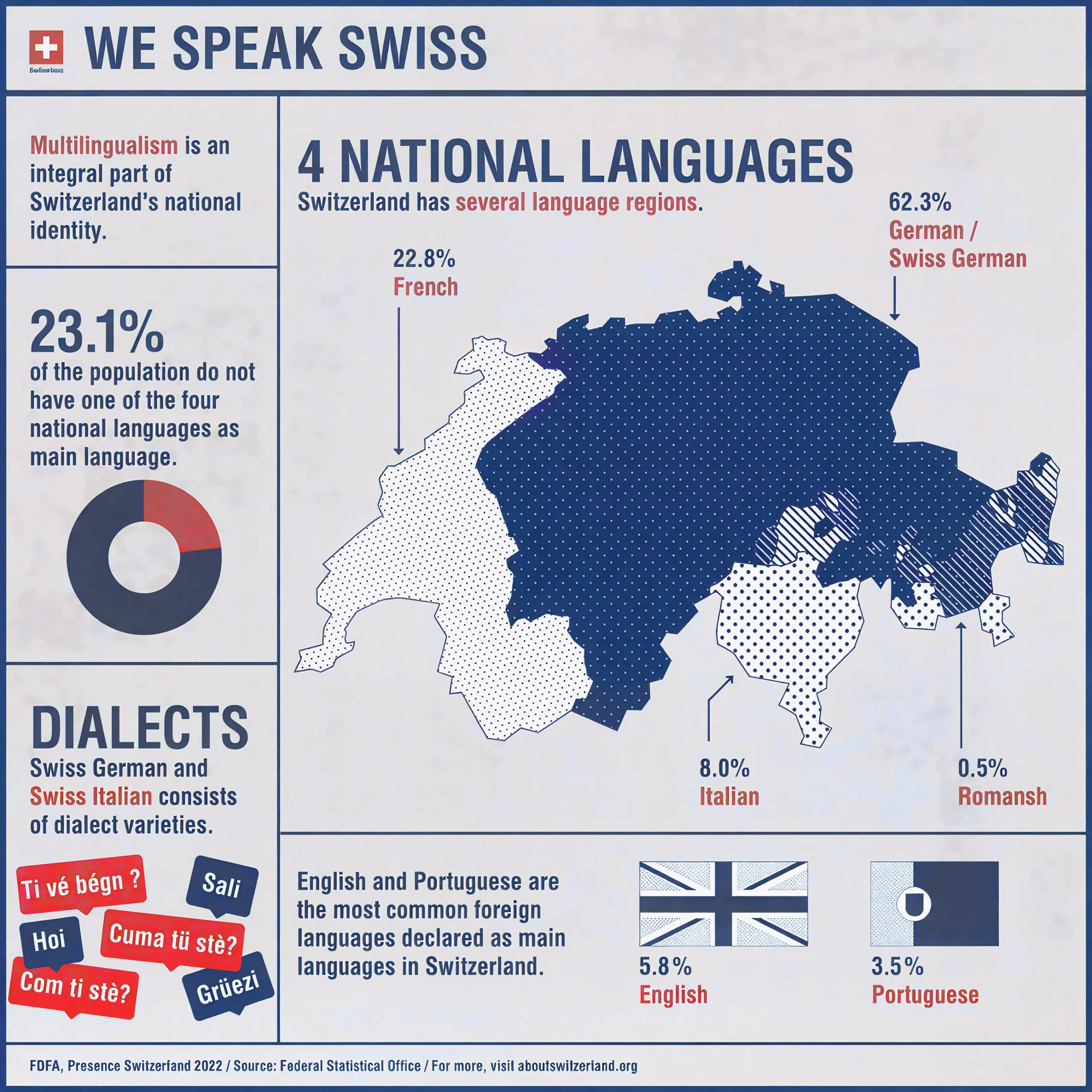
1: German
German is spoken by about 63% of the population and is widely used in northern, central, and eastern Switzerland.
2: French
Approximately 23% of Swiss residents speak French, mainly in the western cantons such as Geneva, Vaud, Neuchâtel, and parts of Bern and Fribourg.
3: Italian
Italian is spoken by around 8% of the population, primarily in Ticino and some areas of Graubünden.
Also Read: Top Languages Spoken In Switzerland
Language and Translation Requirements for Medical Devices in Switzerland
Swissmedic places strong emphasis on language and translation requirements to ensure medical devices are used safely across the country. Under the MedDO, key product information must be available in the official languages so users can clearly understand safety instructions and device details.
Key documents that require translation are:
1: Labeling and Packaging
All labels and packaging must be provided in German, French, and Italian, including essential details such as the device name, batch number, expiry date, and sterilization method.
Recognized international symbols (e.g., ISO 15223-1) may be used to reduce text while maintaining compliance. The Swiss Authorized Representative (CH-REP) and Swiss Importer must also be clearly indicated.
2: Instructions for Use (IFU)
IFUs for devices intended for patients or the general public must always be in all three official languages. For devices used exclusively by healthcare professionals (HCPs), IFUs may be provided in fewer languages or in English, provided the user has the necessary qualifications and patient safety is ensured.
3: Electronic Instructions for Use (eIFU)
eIFUs are digital versions of the IFU, generally permitted only for professional users. They must meet the same language requirements as printed IFUs for HCPs and cannot replace paper IFUs for devices intended for patient use.
4: Certificates & Declarations of Conformity
A: Certificate of Conformity (CoC)
A CoC, issued by an independent certification body, confirms that a device meets regulatory standards. According to Article 25(2) of the MedDO, the CoC must be written in German, French, Italian, or English. If issued in any other language, a translation is required before the device can be marketed in Switzerland.
B: Declaration of Conformity (DoC)
As per Article 29(2) of the MedDO, the DoC must be written in or translated into German, French, Italian, or English, ensuring clear accessibility for Swiss authorities and healthcare professionals.
5: Implant Card
Implant cards are required for long-term implantable medical devices and provide essential information for patients in emergencies or follow-up care.
Under Article 20 of the MedDO, the implant card must always be written in all three official Swiss languages (German, French, and Italian).
No exceptions are permitted due to the critical nature of this information for patient safety and medical response across different regions.
6: Field Safety Notices (FSNs)
Swiss law follows EU MDR Article 89(8), requiring FSNs to be written in the official language(s) of the region where the action occurs.
In Switzerland, this means FSNs must be translated into German, French, or Italian based on the linguistic area affected. This ensures clear, accurate communication during safety incidents and supports quick, effective corrective action.
Looking for medical Translation services?
Language Rules for Patient-Facing vs Professional-Use Devices
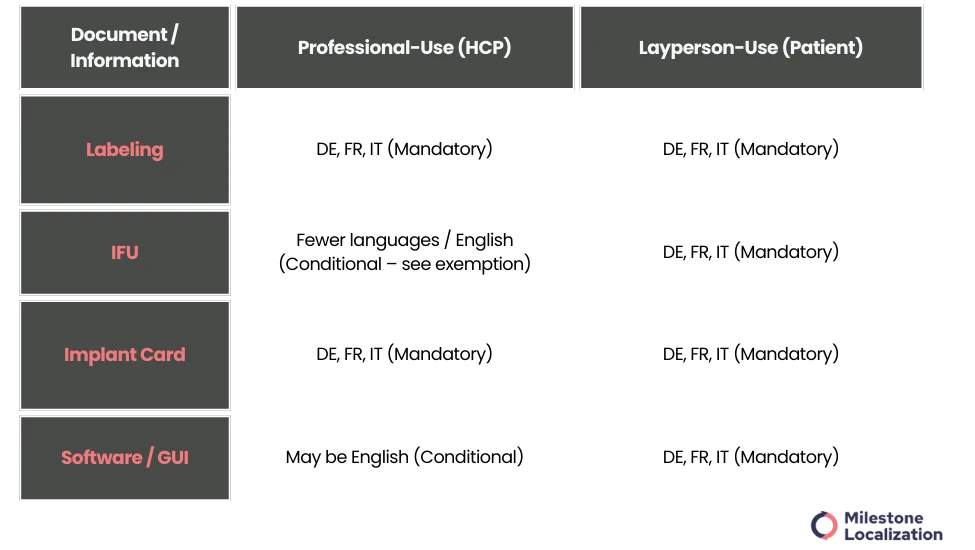
A: Professional-Use Devices (HCPs)
These devices and IVDs are intended exclusively for qualified healthcare professionals in clinical settings (e.g., surgical instruments, lab analyzers).
1: Instructions for Use (IFU)
The IFU may be provided in fewer than the three official languages (DE, FR, IT) or in English, only if all of the following conditions are met:
- The user has the necessary professional and linguistic qualifications to understand the documentation.
- The user agrees to use the device with the limited-language documentation.
- Patient and user safety is not compromised.
- Device performance and clinical efficacy are not impacted by language reduction.
Safety Net: Even if the IFU is provided in English, manufacturers must supply additional information in one of the official languages upon request.
2: Labeling and Packaging
Critical information must still comply with the trilingual rule (DE, FR, IT). Use of internationally recognized symbols is recommended to reduce text while maintaining compliance.
3: Software / GUI
Can be in English if professional users can safely operate the device.
B: Patient / Layperson-Use Devices
These include self-testing, home-use devices, and any documentation directly provided to patients (e.g., glucose meters, hearing aids).
1: Instructions for Use (IFU) & Safety Warnings
Must always be provided in all three official languages (DE, FR, IT) to ensure safe and correct use.
2: Implant Card
Mandatory in DE, FR, IT; carried by the patient for emergencies or follow-up care.
3: Public Summaries (SSCP/SSP)
Must be fully translated into DE, FR, IT.
4: Software / GUI
Must be trilingual to ensure safe operation by patients.
Best practices for medical device translation

1: Work with Certified Medical Translation Agencies
Choose medical device translation agencies that are certified to ISO 17100 (translation quality), ISO 9001 (quality management), and ISO 13485 (medical device quality management) to ensure accuracy, consistency, and regulatory compliance.
These agencies work with professional medical translators and subject matter experts (SMEs) who have deep expertise in medical terminology and device-specific language. They ensure that complex medical content is translated clearly, safely, and in line with Swiss regulatory standards.
Also Read: Top Medical Device Translation Companies
2: Ensure Regulatory Compliance
Strictly adhere to Swiss regulatory requirements under MedDO and IvDO. Translations should accurately convey warnings, instructions, and technical details to ensure full compliance with Swiss legal and safety standards.
3: Use a Standardized Translation Workflow
Implement a collaborative, standardized workflow using translation management systems (TMS), translation memories, and terminology databases. This approach promotes consistent, high-quality translations across all materials.
4: Implement Rigorous Quality Assurance
Establish multi-stage quality assurance processes involving native-speaker linguistic checks, technical reviews by medical experts, and regulatory audits.
5: Documentation & Record Keeping
Maintain detailed records of all translations, checklists, and related materials to accommodate regulatory updates and facilitate ongoing translation management.
6: Align Translation with ISO 13485 Requirements
ISO 13485 defines quality management standards for medical devices, including strict control over documentation.
Aligning your translation processes with ISO 13485 ensures all translated materials are consistent, well-documented, and meet high-quality standards. This helps guarantee accurate, compliant translations that support overall regulatory approval.
Milestone Localization is a Trusted Partner for Medical Device Translations
We help medical device and IVD manufacturers comply with Swissmedic language requirements, ensuring all essential documents are accurately translated into the required languages.
As an ISO 13485, ISO 9001 and ISO 17100 certified company, we ensure that all translations comply with stringent medical device quality management requirements.
We provide translations into German, French and Italian. Our medical translators understand regulatory frameworks, ensuring your documents are accepted by Swiss authorities.
Are You Looking for Certified medical Translation services?
FAQs
What is Swissmedic?
Swissmedic is Switzerland’s regulatory authority responsible for approving, supervising, and ensuring the safety and compliance of medical devices, IVDs, and pharmaceuticals.
What is MEDO (Medical Devices Ordinance)?
MEDO is the Swiss Medical Devices Ordinance that outlines all regulatory requirements for placing medical devices on the Swiss market, including safety, labeling, language rules, and CH-REP obligations.
What is IVDO (In Vitro Diagnostic Devices Ordinance)?
IVDO is the Swiss In Vitro Diagnostic Devices Ordinance that governs the approval and compliance requirements for IVDs in Switzerland, aligned with EU IVDR but adapted to Swiss regulations.
What languages does Swissmedic require for medical device documentation?
Swissmedic requires patient-facing documents to be available in German, French, and Italian. Professional-use devices may use English under specific conditions.
Can Instructions for Use (IFU) be provided only in English in Switzerland?
Yes, but only for professional-use devices and only if Swissmedic’s conditions are met. Lay-user devices must always include DE/FR/IT translations.




