Europe accounts for around 26.4% of the global medical device market. For manufacturers entering the EU, label compliance is one of the practical requirements to get right, and one that is frequently misread.
Many manufacturers approach the physical medical device label the same way they approach the IFU, assuming full translation into every language of every target market is required. That is not what the regulation says.
Here is what the EU MDR actually requires on label language.
What Counts as a “Label” Under EU MDR
EU MDR Article 2(13) defines a label as “the written, printed or graphic information appearing either on the device itself, or on the packaging of each unit or on the packaging of multiple devices.” (EUR-Lex, Regulation (EU) 2017/745)
In practice, this covers three distinct surfaces:
- The device itself, if it is large enough to carry printed information
- The immediate packaging, the packaging in direct contact with the device
- The outer or sales packaging, the box or carton the end user receives
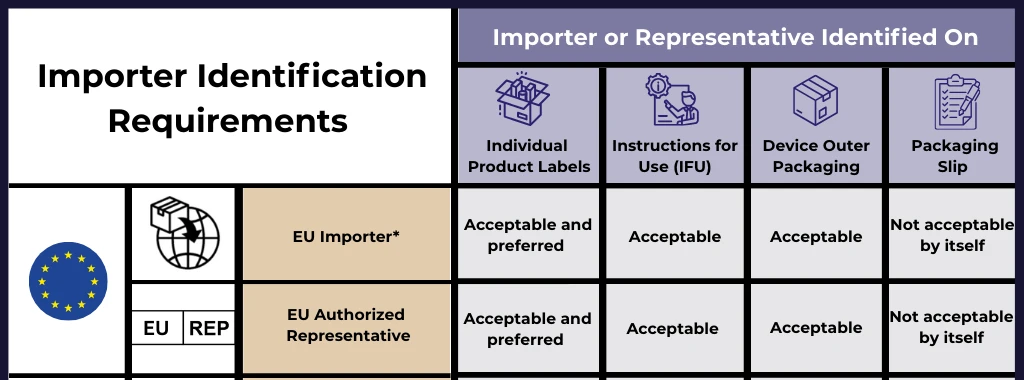
The label is what is physically printed on or attached to the device or its packaging. The requirements for what must appear on labels are set out in Annex I, Chapter III, Section 23.2 of the MDR. (EU MDR Annex I)
Also Read: EU MDR Language Requirements: A Comprehensive Guide
Symbols Do Most of the Work
EU MDR permits the use of internationally recognised symbols in place of written text. The relevant standard is ISO 15223-1:2021. When a recognised symbol is used, no written equivalent is required, and no translation is required for that element.
Symbols under ISO 15223-1:2021 cover a wide range of common label elements: sterility status, single-use designation, expiry, lot number, serial number, manufacturer identification, and date of manufacture, among others.

Several mandatory label elements are also language-neutral by nature and carry no translation obligation:
- UDI (Unique Device Identifier) — numeric or alphanumeric
- Lot and serial numbers — numerical identifiers
- Manufacturing and expiry dates — expressed in year/month format
- CE marking — a standardised graphical mark
- Catalogue or reference numbers — alphanumeric codes
The manufacturer’s name and address, required under Section 23.2, are written in Latin script and are generally accepted as-is across member states.
What Cannot Be Replaced by a Symbol
Symbols cover a significant portion of label content, but not all of it.
Warnings and precautions that go beyond what a standard symbol can communicate must be written out in a language the user can understand.
Intended purpose, when stated on the label, must be comprehensible to the intended user. Article 10(11) requires that information specified in Section 23 of Annex I be provided in an official language determined by the member state where the device is placed on the market.
“Sterile” and “Single Use” have recognised symbol equivalents under ISO 15223-1:2021. However, some member states require a text equivalent alongside the symbol. Requirements for each target market should be verified before assuming the symbol alone is sufficient.
Also Read: IFU Translation: Importance, Requirements & Best Practices
The Multilingual Label: One Label, Multiple Markets
Most manufacturers selling across multiple EU member states do not produce a separate label for each country. They use a single label design that carries all the required languages. This is entirely normal and compliant under EU MDR.
How multilingual labels work in practice
The most common approach is to organise label content into language blocks, each mandatory text element repeated in each required language, stacked in sequence. Some manufacturers use a column format for shorter text strings; others use a running block for longer content.
A widely used system is the language code identifier, sometimes called the flag system. Each language block is prefixed with its two-letter ISO 639-1 code, FR, DE, IT, ES, PT, NL, and so on, so the user can quickly locate their language. This is particularly useful when space is limited and multiple languages appear in proximity.
Managing space constraints
Physical space is a challenge for multilingual labels, especially for small devices. Several solutions are in common use:
- Smaller font sizes, provided legibility requirements are still met (the label must remain “easily legible and clearly comprehensible to the intended user or patient”)
- Folded or booklet labels, which expand the available surface area while keeping the footprint of the packaging unchanged
- Label inserts, a separate printed sheet inside the packaging that carries multilingual content beyond what fits on the external label
The heavier the reliance on symbols, the less text needs to appear in each language, which directly reduces the space required.
Also Read: MDD vs MDR: Key Differences in Translation and Language Requirements
Looking for Medical Device Label Translation services?
Practical Tips for Building Your Multilingual Label
Audit against the ISO 15223-1:2021 symbol library first. Before assigning text to any label element, check whether a recognised symbol exists for it. Every element covered by a symbol is one fewer string to translate across every market and every future label revision.
Map your markets before you translate anything. Identify the specific language requirements for each member state you are selling into. Use the EU Commission’s MDR Language Requirements Table as your starting point. Only translate for confirmed markets.
Build a terminology glossary upfront. Label terminology that needs to stay consistent across markets and across document types. A controlled glossary prevents inconsistencies from developing as label versions are updated.
Design for text expansion. Languages expand when translated from English. German consistently runs longer than its English equivalent. Building this into the label layout from the start avoids legibility issues later.
Treat version control as a compliance requirement. Every label change, revised warnings, updated manufacturer information, new UDI, must trigger a review of all translated versions. A change control process linking label revisions to translation updates is part of maintaining ongoing compliance.
Work with a partner who understands label obligations specifically. Label translation for medical devices is regulatory work. Your translation partner should be familiar with ISO 15223-1:2021, operate within a certified quality management system, and understand that label and IFU obligations are not the same.
Also Read: EUDAMED Translation Requirements for Medical Devices
Conclusion
EU MDR label language requirements are more flexible than many manufacturers expect. Symbols can replace a significant portion of text. A single multilingual design covering multiple markets is standard practice. Codes, numbers, and dates carry no translation obligation.
At the same time, requirements vary by country, some elements must always appear in local language, and the label versus IFU distinction is easy to get wrong. Verifying requirements for each target market before finalising label content avoids compliance issues and delays to market entry.
Milestone Localization is ISO 13485-certified, works across 70+ languages, and has a 100% regulatory acceptance rate for medical device translations.
Get in touch to discuss your label translation requirements → Contact Milestone
Are you looking for certified medical device label translation services?
FAQs
Does EU MDR require labels to be translated into every EU language?
No. EU MDR does not require translation into all 24 official EU languages by default. Article 10(11) requires labels to be in the official language(s) of the specific member states where the device is placed on the market. Manufacturers only need to translate for the markets they are actively selling into.
Can symbols replace text on a medical device label under EU MDR?
Yes. EU MDR permits the use of internationally recognised symbols in place of written text, provided those symbols are drawn from a recognised standard such as ISO 15223-1:2021. When a valid symbol is used, no written equivalent or translation is required.
What is the difference between a label and an IFU under EU MDR?
A label is the written, printed, or graphic information appearing on the device itself or its packaging (Article 2(13)). An IFU is a separate document providing instructions for safe use. The two are distinct under the regulation and carry different language obligations. A common mistake is applying IFU translation rules to the label.
Is the manufacturer's name and address required in local language on an EU label?
No. The manufacturer’s name and address are required under Annex I, Section 23.2, but they are written in Latin script and are generally accepted as-is across EU member states without translation.
How do manufacturers handle multilingual labels for small devices?
Common solutions include folded or booklet labels, label inserts placed inside the packaging, smaller font sizes (provided legibility is maintained), and maximising the use of ISO symbols to reduce the volume of text that requires translation.





