Ever had that sinking feeling when months of hard work hit a roadblock? That’s exactly what happens when companies hear: “Your FDA submission is on hold due to translation issues.”
We’ve seen it firsthand. A promising medical device, a breakthrough drug, or an innovative dietary supplement stuck at the FDA review stage—not because of product problems, but because of preventable translation errors.
The stakes couldn’t be higher. A simple mistranslation can delay your market entry, trigger expensive recalls, or even lead to lawsuits. In this blog, we’ll show you exactly how to navigate FDA translation requirements and protect your investments from these unnecessary risks.
What Is the FDA and Its Oversight Scope?
The Food and Drug Administration stands as America’s regulatory guardian for consumer safety.
Established in 1906, the FDA regulates products accounting for 20 cents of every consumer dollar spent in the United States—approximately $2.6 trillion in consumption annually, according to FDA data.
The FDA’s reach extends beyond U.S. borders to include any product entering American markets. This global oversight makes translation accuracy critical for international companies.
Industries Under FDA Regulation
The FDA’s regulatory authority spans numerous sectors, each with specific translation requirements:
1: Pharmaceuticals and biologics
Clinical trial documentation, drug labeling, adverse event reporting
2: Medical devices
Instructions for use, software interfaces, technical documentation
3: Food and beverages
Nutritional information, allergen warnings, ingredient lists
4: Cosmetics
Safety information, ingredient disclosure, marketing claims
5: Tobacco products
Health warnings, user information, PMTA submissions
6: Dietary supplements
Structure/function claims, ingredient information, safety data
For companies operating across multiple countries, FDA compliance represents just one piece of a complex regulatory puzzle. Navigating these requirements demands specialized knowledge and meticulous attention to detail.
What Are FDA Translation Requirements?
The FDA maintains specific translation requirements across all regulated industries.
All FDA submissions must be in English. Any documents originally created in other languages require certified English translations.
Submission packages must include both the original foreign-language documents and their English translations for verification purposes.
For clinical trials involving non-English speaking participants in the US, patient materials must be translated into the participants’ languages while also providing English versions for FDA review.
Also read: How to Ensure CE Marking Compliance with Accurate Translations – A Guide for Global Businesses
Common Content Requiring FDA-Compliant Translation
The scope of FDA-compliant translation extends to numerous document types:
1: Clinical trial documentation
Protocols, informed consent forms, case report forms
2: GLP study reports
Laboratory findings, methodology descriptions, research outcomes
3: Product labeling and packaging
Usage instructions, warnings, ingredient lists
4: Safety instructions
Proper handling, contraindications, emergency procedures
5: Marketing materials
Claims, promotional content, comparative statements
6: Adverse event reporting
Patient experiences, side effects, unexpected outcomes
7: PMTAs for tobacco products
Product composition, health impact assessments, marketing plans
8: Food compliance documentation
Nutritional facts, allergen information, production standards
Translations must maintain complete fidelity to source documents while adhering to FDA-specific terminology preferences. According to FDA guidance, translator declarations, consistent version control, and traceable processes are essential components of compliant submissions.
The FDA increasingly emphasizes risk-based approaches to documentation. Translation quality should therefore reflect the document’s risk classification—with higher scrutiny for critical safety information.
When Are Translations Mandatory in FDA Submissions?
Know exactly when translations are required to stay compliant.
Not every document requires translation, but knowing which ones do can prevent costly submission delays. The FDA explicitly mandates translations in several key scenarios.
Critical Scenarios Requiring FDA-Compliant Translation
- Clinical trials involving non-English speakers: All participant-facing materials must be translated to ensure informed consent. According to clinical research best practices, translations should undergo linguistic validation to verify conceptual equivalence.
- Foreign-language source documents: Original research, clinical data, or manufacturing information created in another language requires certified translation for FDA review. This applies to international trials, literature references, and collaborative research.
- Product labeling for U.S. markets: All consumer-facing text must appear in English, with precise terminology that meets regulatory standards. The FDA’s Labeling Requirements specify that text must be legible and understandable to the intended user.
- Food and beverage packaging: Imported products must translate all nutritional information, ingredient lists, and allergen warnings. The FDA’s Food Labeling Guide establishes strict formatting and content requirements.
- Structure/function claims: Dietary supplements making health-related claims must ensure accurate translation of all benefit statements. These translations undergo particular scrutiny for potential misrepresentation.
- International adverse event reports: Safety incidents documented in other languages must be translated to support ongoing safety monitoring. Timely and accurate translation is essential for pharmacovigilance.
- Veterinary drug submissions: Animal health products face similar requirements, with translations needed for international data or testing documentation. The Center for Veterinary Medicine maintains specific guidelines.
The FDA applies a risk-based approach—the greater the potential impact on patient or consumer safety, the more stringent the translation requirements become. High-risk medical devices and novel pharmaceuticals face the most rigorous translation scrutiny.
Common Challenges in Meeting FDA Translation Standards
Compliance requires more than language skills—it demands domain expertise and processes.
Even organizations with considerable regulatory experience encounter difficulties when navigating FDA translation requirements. These challenges can delay approvals and increase compliance costs.
1: Technical Terminology and Format Precision
Regulatory documents contain specialized terminology that requires expert knowledge in both languages. Misinterpreting medical, scientific, or legal terms can change meaning in ways that compromise regulatory compliance.
Format requirements add another layer of complexity. The FDA expects specific document structures, heading hierarchies, and terminology conventions. Translations must preserve these elements precisely while maintaining readability.
According to a study in the Journal of Medical Devices, nearly 30% of 510(k) submission delays involve documentation issues, including translation problems.
2: Version Control and Traceability
The FDA expects complete transparency in document evolution. Each translation version must be tracked, with changes documented and justified.
For global companies managing documents across multiple languages, establishing version synchronization presents significant challenges. When source documents change, ensuring all translations remain aligned requires robust systems and workflows.
Linguistic validation—especially for patient-facing materials requires documented evidence that translated content conveys equivalent meaning. This process often involves back-translation, cognitive interviews, and expert review panels.
3: Regulatory Inconsistencies and Legal Implications
FDA requirements sometimes conflict with other international regulations, forcing companies to navigate contradictory expectations. This is particularly challenging for global product launches requiring simultaneous approvals.
Translation errors can have serious legal consequences beyond regulatory rejection. Mistranslated safety information could lead to product liability claims, while inaccurate marketing claims might trigger consumer protection actions.
Companies face the challenge of balancing literal translation against cultural and contextual adaptation. The FDA generally prioritizes literal accuracy, but effective communication sometimes requires cultural sensitivity.
Also read: EU Machinery Directive 2006/42/EC: Translation Requirements
Professional FDA translations?
Who Needs FDA-Compliant Translations?

If your business touches the FDA, you need the right translation partner.
Different industries face unique translation challenges depending on their regulatory pathway and product risk classification. Understanding these distinctions helps target translation resources effectively.
1: Medical Device Manufacturers

Medical device companies require FDA-compliant translation for:
- Instructions for Use (IFUs): Critical safety and operation information for users
- Software interfaces: On-screen text and navigation elements
- Regulatory submissions: 510(k), PMA, and De Novo applications
- Quality system documentation: Procedures, validation reports, and technical files
2: Pharmaceutical and Biotech Companies
The pharmaceutical industry relies on FDA-compliant translation for:
- Clinical trial materials: Protocols, informed consent, patient questionnaires
- Regulatory submissions: INDs, NDAs, and BLAs containing international data
- Pharmacovigilance documents: Adverse event reports and safety updates
- Product labeling: Package inserts, medication guides, patient information
3: Contract Research Organizations (CROs)
CROs conducting multinational research need translation services for:
- Informed consent forms: Ensuring participants understand study details
- Case report forms: Standardizing data collection across languages
- Investigator brochures: Communicating product information to research sites
- Clinical study reports: Synthesizing findings for regulatory submission
Also read: The Role of CROs in Managing Multilingual Clinical Trial Documentation
4: Food and Beverage Manufacturers
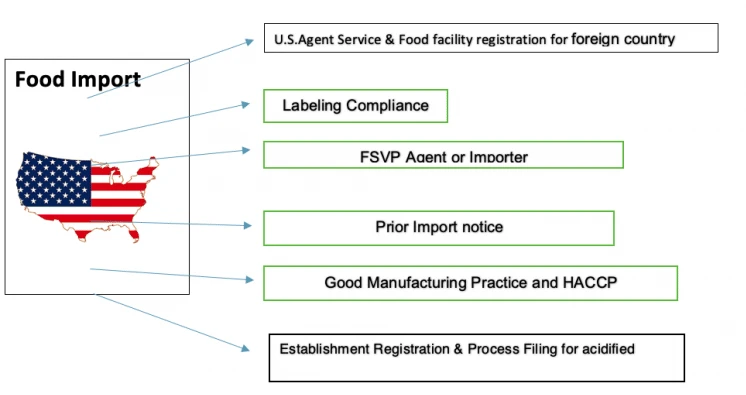
Companies importing food products to the U.S. require translation for:
- Nutritional labeling: Caloric content, serving sizes, daily value percentages
- Ingredient lists: Complete component disclosure following U.S. conventions
- Allergen warnings: Clear identification of major allergens
- Health claims: Properly qualified statements about nutritional benefits
5: Cosmetic and Personal Care Product Companies
Cosmetics manufacturers need translation support for:
- Ingredient disclosures: INCI name listings and concentrations
- Safety information: Usage warnings and precautions
- Marketing claims: Carefully calibrated benefit statements
- Manufacturing documentation: GMP compliance evidence
6: Tobacco and E-Cigarette Companies
The tobacco industry faces rigorous translation requirements for:
- Premarket Tobacco Applications: Comprehensive product documentation
- Health warnings: Mandatory caution statements
- User instructions: Proper usage directions
- Marketing materials: Age-restricted promotional content
7: Dietary Supplement Companies
Supplement manufacturers require FDA-compliant translation for:

- Structure/function claims: Carefully worded benefit statements
- Supplement Facts panels: Standardized nutrient information
- Warning statements: Required precautionary language
- New dietary ingredient notifications: Safety substantiation documentation
8: Veterinary Product Manufacturers
Animal health companies need translation services for:
- Product labeling: Usage directions for veterinarians and pet owners
- Safety studies: Research documentation supporting approval
- Manufacturing information: Production standards and quality controls
- Post-market surveillance: Adverse event reporting from international markets
Our Process: How We Deliver FDA-Ready Translations
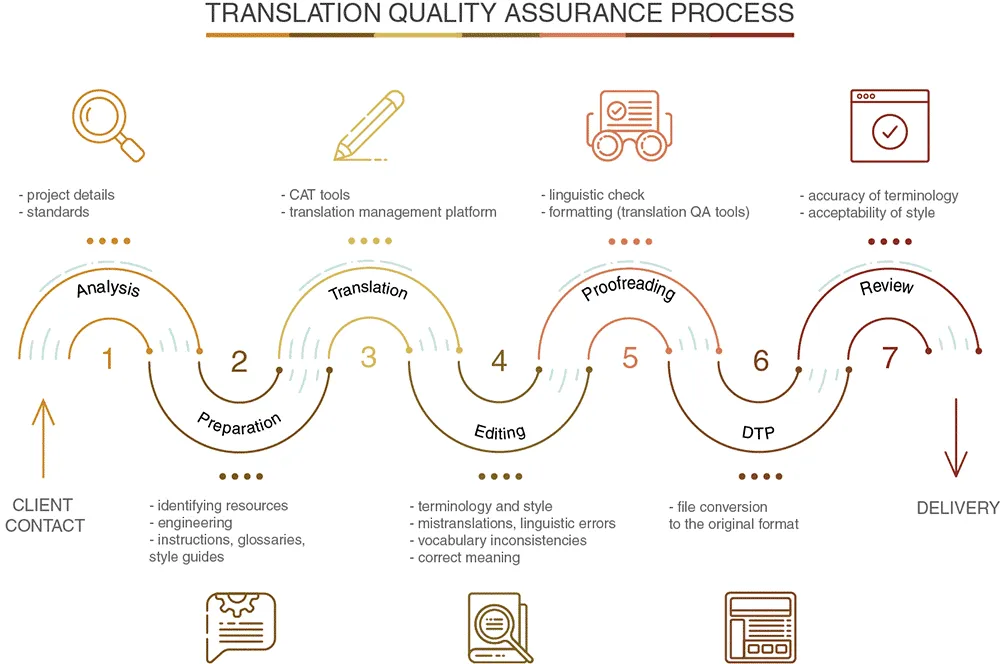
A proven, repeatable, auditable translation workflow built for FDA requirements.
Meeting FDA translation standards demands more than linguistic expertise—it requires systematic processes designed specifically for regulatory compliance. Our methodology ensures translations maintain both accuracy and regulatory alignment.
1: The T+P Model for Quality Assurance
Our core approach follows the T+P (Translation+Proofreading) model, considered the gold standard for regulatory documentation:
- Translation by subject matter experts: Our translators possess both linguistic fluency and domain credentials in your specific industry
- Proofreading by senior linguists: Specialized editors review terminology precision, formatting consistency, and regulatory alignment
- Review by quality assurance specialists: Final verification ensures perfect accuracy before delivery
This multi-layer approach reduces error risk by applying different expertise at each stage.
2: Compliance Documentation and Validation
Regulatory submissions require evidence of translation validity. Our process includes:
- Translator declarations: Formal certification of translation accuracy by qualified professionals
- Back translation: Independent reverse translation to verify meaning preservation for clinical trial documents that mandate it
- Audit documentation: Comprehensive records of all process steps, reviewer qualifications, and quality checks
3: Technology-Enhanced Quality Control
Our translation process leverages specialized tools that enhance both quality and consistency:
- CAT (Computer-Assisted Translation) tools: Maintain terminology consistency and leverage approved content
- Version tracking systems: Document all changes with timestamps and responsible parties
- Term bases and translation memories: Ensure regulatory-specific terminology remains consistent across all documents
Why Our ISO 17100 & ISO 9001 Certifications Matter for FDA Compliance
Certifications aren’t badges—they’re your guarantee of accuracy, quality, and process integrity.
While the FDA doesn’t explicitly require ISO certification for medical device translation providers, these international standards align perfectly with regulatory expectations. Our certifications demonstrate our commitment to quality processes that support compliance.
1: ISO 17100: The Translation Quality Standard
ISO 17100 specifically addresses translation service quality through:
- Translator qualification requirements: Verified education, experience, and subject matter expertise
- Mandatory review processes: Independent checking of all translated content
- Documentation standards: Comprehensive record-keeping of all translation activities
2: ISO 9001: Quality Management Excellence
Our ISO 9001 certification extends quality assurance beyond translation to encompass all organizational processes:
- Consistent client communication: Structured requirements gathering and specification development
- Continuous improvement: Regular process evaluation and enhancement based on outcomes
Risk management: Proactive identification and mitigation of potential quality issues
Conclusion
The difference between regulatory success and costly delays often comes down to translation quality.
Forward-thinking companies treat FDA-compliant translation as a strategic investment rather than a box to check. They integrate specialized translation partners into their development process from the beginning.
This strategic approach doesn’t just satisfy regulators—it accelerates approvals, reduces costs, and ultimately delivers innovations to market faster than competitors. In today’s global marketplace, that’s not just good compliance—it’s good business.
About Milestone Localization
Milestone Localization specializes in FDA-compliant translation services for regulated industries. Our ISO 17100 and 9001 certified processes ensure your documentation meets the highest standards of accuracy and regulatory alignment.
Our team includes subject matter experts in pharmaceuticals, medical devices, food and beverages, cosmetics, and dietary supplements. This specialized knowledge ensures translations maintain both technical precision and compliance with FDA terminology requirements.
Contact us today to discuss your specific FDA translation needs and discover how our certified processes can streamline your regulatory submissions.
Translations in compliance with FDA requirements
FAQS ON FDA Translation Requirements
What are the FDA translation requirements for products entering the US market?
All FDA submissions must be in English. Documents originally in other languages require certified English translations, and both the original and English versions must be submitted for verification.
Which types of documents require FDA-compliant translation?
Common documents include clinical trial protocols, informed consent forms, product labeling and packaging, safety instructions, marketing materials, adverse event reports, and more depending on the industry sector.
Does the FDA mandate translation of medical device instructions and labels?
The FDA requires that device labels and instructions be understandable by the intended audience. While not all devices need translations, those marketed to populations with limited English proficiency may require them.
Are there any specific terminologies or standards translators must follow for FDA submissions?
Yes, translations must maintain complete fidelity to the source documents and adhere to FDA-preferred terminology. Translator declarations, version control, and traceable processes are essential.
Is translation required for clinical trial documents involving non-English speaking participants in the US?
Yes, patient materials must be translated into the participants’ languages alongside English versions for FDA review to ensure transparency and compliance.
Are certified translations required for FDA document translations?
Certified translations by native speakers with domain expertise are recommended to meet FDA standards and avoid errors in regulatory submissions.
Do FDA translation requirements vary across industries?
Yes, industries under FDA regulation such as pharmaceuticals, medical devices, food and beverages, cosmetics, tobacco products, and dietary supplements have specific translation needs tailored to their regulatory documents.
Can a translation be updated or corrected after submission to the FDA?
Yes, corrections or updates must be promptly submitted. Clearly reference the revised document versions and provide reasons for the changes.
How frequently do FDA translation regulations change, and how should companies stay informed?
FDA guidance and regulations can evolve. Stay informed through the FDA website, regulatory updates, and professional newsletters, and consider consulting regulatory experts for major changes.

